 Today’s approval is an important advance for these patients as it allows them to benefit from triple therapy by using one inhaler, once-a-day.” Today’s announcement marks GSK’s sixth major medicine approval in 2020 across areas of significant unmet medical need including cancer, HIV, respiratory and chronic kidney disease.ĭr Hal Barron, Chief Scientific Officer and President R&D, GSK, said: “Millions of asthma patients in the US rely on multiple inhalers to help control their condition and manage their symptoms. The approval means Trelegy is the first single inhaler triple therapy approved for the maintenance treatment of both asthma and COPD and is the only single inhaler triple therapy available for patients in a convenient once-daily inhalation in the US. 
There is an additional strength for asthma alone which is fluticasone furoate / umeclidinium / vilanterol 200/62.5/25mcg. The FDA-approved strength for both COPD and asthma is fluticasone furoate / umeclidinium / vilanterol 100/62.5/25mcg. 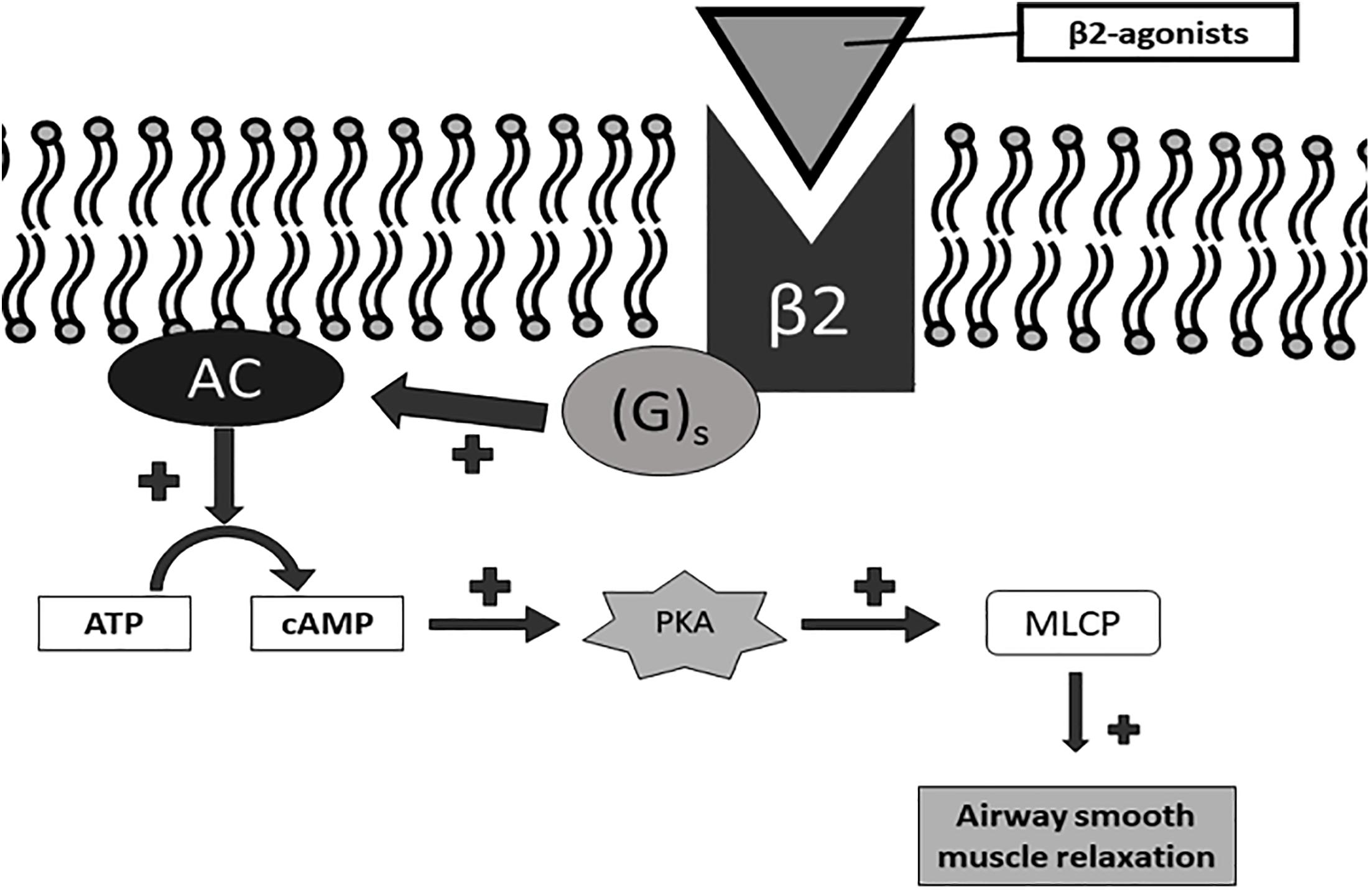
Trelegy Ellipta is not indicated for relief of acute bronchospasm. (NASDAQ: INVA) announced the US Food and Drug Administration (FDA) has approved a new indication for Trelegy Ellipta (fluticasone furoate / umeclidinium / vilanterol ‘FF/UMEC/VI’) for the treatment of asthma in patients aged 18 years and older adding to its current license for use in patients with chronic obstructive pulmonary disease (COPD). GlaxoSmithKline plc (LSE/NYSE: GSK) and Innoviva, Inc. Issued: 9 September 2020, London UK New asthma indication for Trelegy Ellipta introduces an important option for patients to the current treatment paradigm 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
